Troubleshooting: Why Is Your SLES Not Thickening Properly or Turning Hazy?
Have you ever formulated a shampoo or body wash that simply refuses to thicken—no matter how much salt you add? Or watched your once crystal-clear liquid turn hazy and unappealing after production? These frustrating problems affect product quality, waste valuable materials, and can even force you to reject entire batches.
As a manufacturer of SLES (Sodium Laureth Sulfate), we regularly hear from customers battling thickening failures and clarity issues. The good news? Both problems are solvable. They typically trace back to a few key factors: improper salt usage, SLES quality variations, pH imbalances, or ingredient interactions.
In this article, we’ll explore exactly why SLES formulations fail to thicken or turn hazy—and provide a practical 7-step checklist to help you fix these issues for good.
Problem 1: Why Your SLES Won’t Thicken
The “Salt Curve” Explained
SLES thickening follows what formulators call a “salt curve”—imagine climbing a hill:
Add salt, viscosity rises
Hit the peak (sweet spot) for maximum thickness
Add too much salt, and viscosity crashes
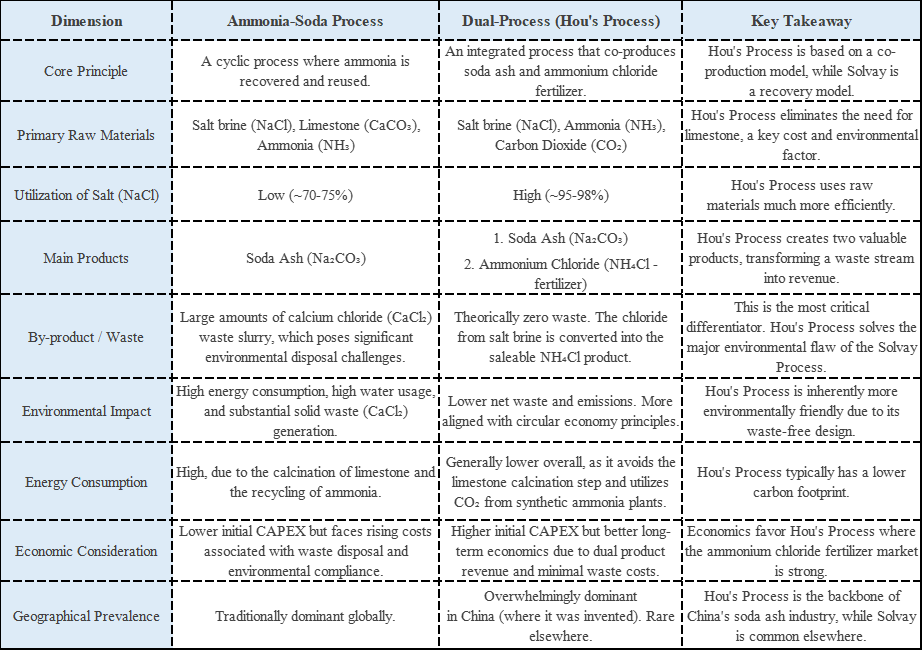
Here’s the science: surfactant molecules form tiny structures called “micelles” in water. The right amount of salt makes these micelles pack tightly together, creating networks that trap water and build viscosity. But excess salt destroys these networks, breaking micelles apart and leaving you with thin, watery products.
Every formula has a unique peak, typically between 1-4% salt. Change your SLES supplier, add fragrance, or modify other ingredients—and that peak shifts.
SLES Quality Matters: The Free Alcohol Problem
Lower-quality SLES contains more unreacted fatty alcohols (measured as “unsulfated matter”). These impurities interfere with micelle formation, preventing proper thickening regardless of how much salt you add.
We’ve seen this firsthand: a formulator whose recipe worked perfectly for months suddenly faced hazy, thin products after switching SLES batches. The culprit? Unsulfated matter jumped from 0.7% to 1.8%.

pH and Temperature Effects
pH matters. SLES systems thicken best at pH 5.5-6.5. Outside this range, micelle structures change and viscosity drops.
Temperature matters too. That perfectly thick shampoo at 25°C may gel in cold weather or turn watery in tropical heat. Salt makes cold-temperature gelling worse—not better.
Ingredient Interference
Watch for formulation “thieves” that steal your viscosity:
PEG-40 Hydrogenated Castor Oil (common solubilizer) kills both thickness and foam
High fragrance levels (over 1%) disrupt thickening
Certain cationic polymers interfere with salt response
Problem 2: Why Your Clear Products Turn Hazy
Salt Overload = “Salting Out”
Adding too much salt doesn’t just ruin viscosity—it forces surfactants to separate from water. This “salting out” creates an unappealing white, hazy appearance that’s nearly impossible to reverse.

SLES Quality and Clarity
Those same free alcohols that hurt thickening also cause haze. They precipitate at cooler temperatures, making your product look cloudy. This is why low-unsulfated SLES is essential for crystal-clear formulations.
EO Degree Choice
SLES comes in different ethoxylation grades. Research shows SLES-3EO (3 moles ethylene oxide) tends to produce higher haze than SLES-2EO. If clarity is your priority, choose your SLES type carefully.
Other Clarity Killers
Cationic polymers (in conditioning shampoos) increase haze at higher levels
Insufficient viscosity lets pearlescent agents settle out
Cold storage forces surfactant components to precipitate
7-Step Troubleshooting Checklist
When your formulation misbehaves, work through these steps systematically
Step 1: Run a Salt Curve Test
Find your formula’s peak:
Prepare 500g of base (no thickener)
Add salt in 0.5% increments
Measure viscosity after each addition
Identify the peak—then back off slightly for production safety

Pro tip: Never dump dry salt directly into your batch. Make a 20-25% salt solution and add gradually for better control.
Step 2: Check Your Chelating Agent
Hard water minerals (calcium, magnesium) interfere with micelles. Ensure 0.1-0.2% EDTA (or equivalent) is present. Increase if your water is very hard.
Step 3: Verify and Adjust pH
Target pH 5.5-6.5. Use citric acid to lower pH, sodium hydroxide to raise it. Adjust pH first, then fine-tune salt—never the reverse.
Step 4: Confirm SLES Quality
Review your Certificate of Analysis (COA):
Active matter: 68-72% (for 70% grade) or 27-29% (for 28% grade)
Unsulfated matter: Should be low—ideally below 1.0%
Color (APHA): ≤30 indicates good quality
If you suspect batch variation, request multiple samples for comparison testing.
Step 5: Evaluate Formulation Compatibility
Scan your ingredient list for known interference:
Solubilizers like PEG-40 hydrogenated castor oil
Fragrance above 1%
Cationic polymersSometimes swapping one ingredient fixes everything.
Step 6: Temperature Stability Testing
Test your product under real-world conditions:
Room temperature (25°C) — 24 hours
Tropical storage (45°C) — 48 hours
Cold transport (4°C or ice bath) — 24 hours
Freeze-thaw cycles (-5°C to 25°C, 3 cycles)
Note any haze, gelation, or separation.
Step 7: Consider Co-thickeners
If salt alone won’t get you there:
CMEA (1-3%): Boosts foam and helps thickening
CAPB (3-8%): Adds mildness and aids viscosity
Cellulosics or Carbomer: For high-viscosity needs (higher cost, requires neutralization)
Case Study: Real Problem, Real Solution
The Situation: A mid-sized manufacturer produced crystal-clear body wash for years using the same formula. After switching SLES suppliers, the product turned hazy and lost 30% of its viscosity.
The Investigation:
Salt curve shifted from 2.5% peak to 1.8% peak
New SLES showed 1.8% unsulfated matter (old batch: 0.7%)
pH measured 6.8—above optimal range

The Fix:
Reduced salt from 2.3% to 1.6%
Adjusted pH to 6.2
Added 0.2% EDTA for stabilityThe Result: Product clarity returned, viscosity targets were met, production resumed normally.
How Quality SLES Prevents These Problems
At YRC, we’ve spent decades helping formulators avoid these exact headaches. Our SLES offers:
Low unsulfated matter (<0.8%) — fewer impurities means better thickening and clarity
Consistent batch quality — key parameters vary less than 5% between lots
Multiple EO options — choose SLES-2EO or SLES-3EO for your specific clarity needs
Expert technical support — our formulators (average 10+ years experience) are here to help

Three Rules to Remember
Salt is the cheapest thickener—and the most dangerous. Always find your peak in the lab and leave a safety margin in production.
Your SLES quality sets your formulation ceiling. Choose suppliers who deliver consistent, low-unsulfated material.
Systematic beats random. When problems appear, work through the checklist—don’t just add “a little more salt.”
Conclusion
Thickening difficulties and clarity issues are common challenges in formulation development, but they’re not insurmountable. By understanding the “salt curve,” choosing high-quality SLES, strictly controlling pH, and systematically optimizing your formulation, you can consistently produce high-quality products with ideal viscosity and crystal clarity.

Never add “just a little more salt” on the production line without testing—once the product turns thin and hazy, it’s almost impossible to fix.
Facing a stubborn formulation problem? Our technical team offers free consultation. Contact us for expert help.